Classical semi-quantitative and quantitative capillaroscopy interpretation
This bridge unit organizes the classical reading of nailfold capillaroscopy into three complementary levels: recognizing whether the pattern is normal, nonspecific, or scleroderma-like; grading the main findings using semi-quantitative logic; and measuring the variables that remain most robust in clinical practice, especially capillary density [1, 2, 3, 4].
- The most useful sequence remains to read density first, then dilations or giant capillaries, hemorrhages, and finally architectural disorganization and neoangiogenesis [2, 3, 4].
- Classical semi-quantification reduces part of the subjectivity, but it only works if acquisition is standardized and the reader uses reproducible definitions [2, 4, 5, 6].
- Capillary density is the most stable classical quantitative metric and the one that best helps separate nonspecific variation from relevant microangiopathy [2, 3, 4].
- The Maricq and Cutolo patterns remain very useful for teaching and for systemic sclerosis, although they simplify a more heterogeneous clinical reality [4, 5, 6, 7].
- Current practice does not replace classical frameworks: it integrates them with structured reporting, uniform terminology, and, when possible, automated analysis [2, 3, 4, 7, 8, 9].
Learning path
This unit fits after nailfold microcirculation, because it turns anatomy and physiology into a practical reading sequence. Afterward, it will be easier to review equipment and devices and acquisition and quality to understand the technical conditions needed for reliable measurement, and later to connect with normality and variability and semiology and reporting.
From clinical eye to reproducible data
Classical interpretation is not about memorizing a list of isolated findings. It is about moving from a global impression to an ordered description and, when useful, to measurements that can be compared between visits or readers.
In teaching and clinical practice, it is helpful to distinguish three complementary levels. The descriptive level decides whether the image is normal, nonspecific, or compatible with a scleroderma pattern. The semi-quantitative level grades the magnitude of the main abnormalities using simple categories. The quantitative level measures variables such as density or diameters, which best withstand longitudinal comparison [2, 3, 4].
| Level | Main question | What it adds | Main limitation |
|---|---|---|---|
| Descriptive | Is the pattern normal, nonspecific, or scleroderma-like? | Allows rapid classification and guides clinical suspicion [2, 4, 7] | May be insufficient for fine follow-up |
| Semi-quantitative | How much relative involvement is present in each domain? | Reduces subjectivity and orders severity [4, 5, 6] | Strongly depends on definitions and sampling |
| Quantitative | Which variables can I measure with reasonable reproducibility? | Improves follow-up, research, and automation [2, 3, 4, 8, 9] | Not every morphological finding is equally robust to measure |
The most useful practical sequence remains to read in this order: capillary density, dilations or giant capillaries, hemorrhages, architectural disorganization, and finally avascular areas and neoangiogenesis. This sequence forces the interpretation of combinations of findings rather than isolated lesions [2, 3, 4, 7].
Classical semi-quantitative interpretation
Classical semi-quantification attempted to turn visual observation into a language of severity. It remains useful for teaching how to read and for comparing studies when a complete automated system is not available.
The classical approach analyzes a central strip of the nailfold bed and translates the relative frequency of each finding into simple categories. Historically, many groups counted the percentage of capillaries affected in each domain and assigned 1 if involvement was below 33%, 2 if it was between 33% and 66%, and 3 if it exceeded 66% [5, 6]. In teaching, that logic is often simplified to a 0-to-3 scale for each domain, always using standardized images and several fingers.
| Domain | What is assessed | Why it helps | Risk if misused |
|---|---|---|---|
| Dilations or giant capillaries | Relative frequency of homogeneously dilated loops | Helps recognize early or active scleroderma pattern [4, 5, 6, 7] | Overcalling mild ectasias or focus artifacts |
| Microhemorrhages | Number and distribution of recent or residual hemorrhages | Adds weight when it coexists with giant capillaries [4, 5, 6, 7] | Confusing microtrauma with microvascular damage |
| Density loss | Relative absence of distal loops | Orders structural severity and follow-up [2, 3, 4, 5, 6] | Applying rigid thresholds without technical or biological context |
| Neoangiogenesis or abnormal shapes | Ramifications, bushy shapes, or reparative architecture | Suggests advanced damage when it coexists with capillary loss [4, 5, 6, 7] | High inter-reader variability |
Its strength is pedagogical: it forces the reader to think in percentages, domains, and relative severity. Its weakness is equally clear: a field that is too narrow, a suboptimal image, or selection of too few fingers can substantially distort the final score [2, 3, 4, 6]. A 2026 multicenter study made this visible at the classification level: reducing full 32-image coverage of the eight non-thumb fingers led to frequent shifts between systemic sclerosis and non-systemic sclerosis patterns and between Cutolo subtypes [10]. For that reason, isolated semi-quantification is now used less as the sole basis for a clinical report and more as support for structured reading.
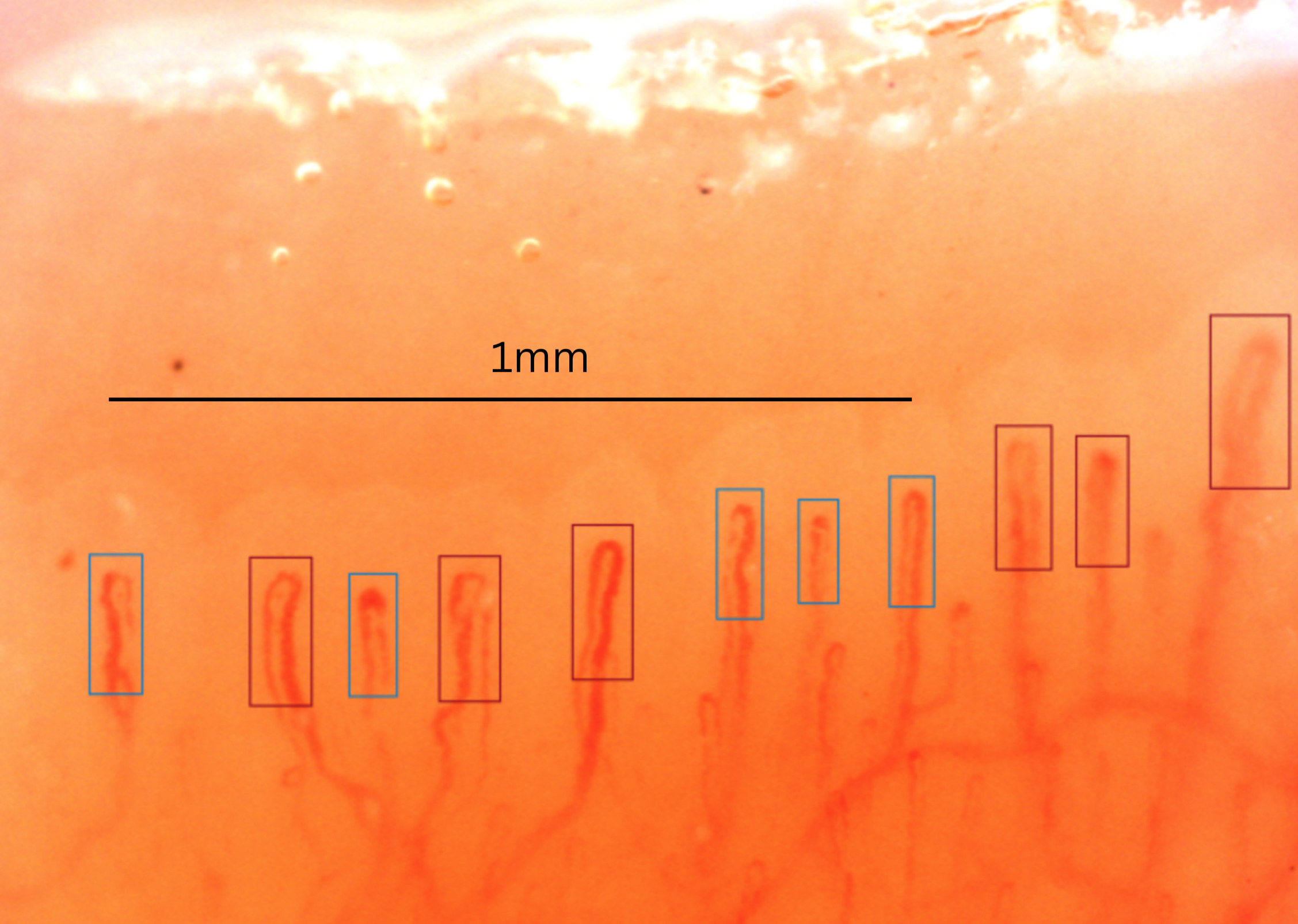
Classical quantitative interpretation
Classical quantification tries to measure what can truly be compared between visits: how many capillaries are present, how large they are, and how extensive structural loss is. Not all metrics have the same practical utility.
In daily practice, capillary density is the core of quantitative analysis because it is the most robust variable and the one that best integrates normality, progression, and follow-up [2, 3, 4]. Diameters, percentage of giant capillaries, and extent of avascular areas are also useful, but they are more sensitive to focus, calibration, and software. Modern automation aims precisely to enlarge the analyzed surface and reduce inter-reader variability [3, 8, 9].
| Metric | How it is expressed | Main utility | Limitation |
|---|---|---|---|
| Capillary density | Capillaries/mm in the distal row | Discriminates significant microangiopathy and helps follow-up [2, 3, 4] | Varies with technique, selected finger, and image quality |
| Apical diameter | Micrometers | Helps separate ectasia, marked dilation, and giant capillary [2, 3, 4] | Requires reliable focus and calibration |
| Giant capillaries | Number or percentage | Reinforces recognition of the scleroderma pattern [4, 5, 6, 7] | May decrease in late phases despite more advanced damage |
| Avascular areas | Presence, number, or extent | Express relevant microvascular loss [4, 5, 6, 7] | Operational definitions vary between series |
| Neoangiogenesis | Presence or grade | Suggests disorganized repair and advanced damage [4, 5, 6, 7] | Its reproducibility remains lower |
Strongest evidence: capillary density and combinations of structural findings. Intermediate evidence: diameter measurements and composite scores. Emerging evidence: large-scale automated quantification of the nailfold bed [2, 3, 4, 8, 9].
Classical Maricq and Cutolo patterns
Classical classifications were decisive because they taught capillaroscopy as an evolving process. Their value is not that they are perfect, but that they force the reader to recognize coherent configurations of microvascular damage.
Maricq: a conceptual entry point
The classical reading attributed to Maricq distinguishes a slow pattern, dominated by dilations and megacapillaries with mild capillary loss, from an active pattern, in which capillary loss and vascular disorganization carry more weight [4, 5]. Its teaching value remains clear: it shows that capillaroscopy is not a sum of isolated findings, but a configuration of microangiopathy with different degrees of destruction.
| Pattern | Dominant findings | Practical reading |
|---|---|---|
| Maricq slow | Dilations and megacapillaries, with architecture still relatively preserved | Less destructive scleroderma-spectrum microangiopathy [4, 5] |
| Maricq active | More intense capillary loss and evident disorganization | More advanced damage, with less relative weight from giant dilations [4, 5] |
Cutolo: early, active, and late
Cutolo refined that sequence and proposed the three patterns that remain the most widely used in systemic sclerosis: early, active, and late [4, 5, 6, 7]. The logic is evolutionary: first, megacapillaries and hemorrhages predominate with little capillary loss; then moderate loss and disorganization appear; finally, avascular areas and neoangiogenesis dominate, with fewer typical megacapillaries visible.
| Cutolo pattern | What predominates | What to remember |
|---|---|---|
| Early | A few megacapillaries and some hemorrhages, with relatively preserved architecture | Capillary loss is minimal or absent [4, 5, 6, 7] |
| Active | Frequent megacapillaries and hemorrhages, with moderate loss and initial disorganization | This is the "classic" pattern of intermediate progression [4, 5, 6, 7] |
| Late | Severe capillary loss, avascular areas, and neoangiogenesis | There may be fewer visible megacapillaries despite greater overall severity [4, 5, 6, 7] |
Limitations and current relevance
Maricq and Cutolo remain essential for understanding the natural history of the scleroderma pattern, but today they should be taught and interpreted within a stricter methodological framework.
- They do not cover all interindividual variability or all biological heterogeneity in systemic sclerosis [4, 7].
- They simplify a progression that is not always linear or unidirectional [4, 7].
- They depend on acquisition quality and reader expertise; reproducibility falls outside expert settings [2, 3, 4].
- They perform less well when nonspecific findings or diseases other than systemic sclerosis predominate [2, 4, 7].
- They do not replace structured reporting or objective quantification of robust domains [2, 3, 4].
- A single examination may be insufficient to classify very early phases or mixed findings with confidence [2, 4, 7].
The current message is not to abandon classical classifications, but to integrate them with standardized acquisition, uniform terminology, minimum reporting domains, and increasingly broad quantitative analysis. Automated systems promise to reduce variability, but they still depend on good image quality and an appropriate clinical framework [2, 3, 4, 7, 8, 9].
Reading checklist
- Confirm that the distal row is assessable and that there is no pressure-induced collapse.
- Review capillary density across several fingers before focusing on one striking isolated finding [2, 3, 4].
- Distinguish mild dilations from homogeneous and repeated giant capillaries [2, 4].
- Assess whether hemorrhages are plausibly traumatic or part of a structured pattern [2, 4, 7].
- Document capillary loss, avascular areas, and neoangiogenesis using stable language [2, 3, 4].
- If a semi-quantitative score is used, state clearly how many fingers and what surface area were analyzed [5, 6, 10].
- Do not conclude a classical pattern without integrating technique, clinical context, and temporal evolution [2, 3, 4, 7].
FAQ
Do semi-quantitative and quantitative mean the same thing?
No. Semi-quantification groups severity into simple categories; quantification attempts to measure continuous or ordinal variables such as density or diameters [2, 3, 4].
Which classical metric deserves the most confidence in clinical practice?
Capillary density remains the most robust quantitative variable, provided the image is assessable and the count is performed using a standardized technique [2, 3, 4].
Is a late pattern with few megacapillaries less severe?
No. In Cutolo's classification, reduction in megacapillaries may coexist with greater overall severity because capillary loss and neoangiogenesis dominate [4, 5, 6, 7].
Can I use a classical scale on a single image?
It is not good practice. The score loses value if it is not based on several fingers and on images acquired in a comparable way [2, 3, 4, 6].
Are the Maricq and Cutolo patterns useful outside systemic sclerosis?
They are mainly useful as a conceptual framework and perform best within the scleroderma spectrum. Outside that context, specificity decreases [2, 4, 7].
Glossary
- Descriptive interpretation
- Initial reading that determines whether capillaroscopy is normal, nonspecific, or compatible with a scleroderma pattern.
- Semi-quantification
- Grading of capillaroscopic findings into simple severity categories, typically from 0 to 3 or by percentage intervals.
- Quantification
- Measurement of reproducible variables such as capillary density or diameters, with the aim of comparing studies over time or between readers.
- Giant capillary
- A homogeneously dilated capillary; it corresponds to the historical term megacapillary and carries particular weight within the scleroderma pattern.
- Avascular area
- Area of the distal row with relevant loss of visible capillaries, indicating microvascular destruction when the technical context is appropriate.
- Neoangiogenesis
- Formation of vessels with abnormal or reparative appearance, typical of more advanced phases of microvascular damage.
- Late pattern
- Advanced phase in Cutolo's classification, characterized by intense capillary loss, avascular areas, and neoangiogenesis.
References
- Gracia Tello B, Ramos Ibañez E. Nailfold capillaroscopy. Med Clin (Barc). 2023;160(11):499-500. doi: 10.1016/j.medcli.2023.01.011. PMID: 36907714.
- Smith V, Herrick AL, Ingegnoli F, Damjanov N, De Angelis R, Denton CP, et al. Standardisation of nailfold capillaroscopy for the assessment of patients with Raynaud's phenomenon and systemic sclerosis. Autoimmun Rev. 2020;19(3):102458. doi: 10.1016/j.autrev.2020.102458. PMID: 31927087.
- Herrick AL, Berks M, Taylor CJ. Quantitative nailfold capillaroscopy-update and possible next steps. Rheumatology (Oxford). 2021;60(5):2054-2065. doi: 10.1093/rheumatology/keab006. PMID: 33493310.
- Smith V, Ickinger C, Hysa E, Snow M, Frech T, Sulli A, et al. Nailfold capillaroscopy. Best Pract Res Clin Rheumatol. 2023;37(1):101849. doi: 10.1016/j.berh.2023.101849. PMID: 37419757.
- Cutolo M, Sulli A, Pizzorni C, Accardo S. Nailfold videocapillaroscopy assessment of microvascular damage in systemic sclerosis. J Rheumatol. 2000;27(1):155-160. PMID: 10648032.
- Sulli A, Secchi ME, Pizzorni C, Cutolo M. Scoring the nailfold microvascular changes during the capillaroscopic analysis in systemic sclerosis patients. Ann Rheum Dis. 2008;67(6):885-887. doi: 10.1136/ard.2007.079756. PMID: 18037628.
- Smith V, Vanhaecke A, Herrick AL, Distler O, Guerra MG, Denton CP, et al. Fast track algorithm: How to differentiate a "scleroderma pattern" from a "non-scleroderma pattern". Autoimmun Rev. 2019;18(11):102394. doi: 10.1016/j.autrev.2019.102394. PMID: 31520797.
- Bharathi PG, Berks M, Dinsdale G, Murray A, Manning J, Wilkinson S, et al. A deep learning system for quantitative assessment of microvascular abnormalities in nailfold capillary images. Rheumatology (Oxford). 2023;62(6):2325-2329. doi: 10.1093/rheumatology/kead026. PMID: 36651676.
- Ebadi Jalal M, Emam OS, Castillo-Olea C, García-Zapirain B, Elmaghraby A. Abnormality detection in nailfold capillary images using deep learning with EfficientNet and cascade transfer learning. Sci Rep. 2025;15(1):2068. doi: 10.1038/s41598-025-85277-8. PMID: 39814806.
- Guillén del Castillo A, Lledó-Ibáñez GM, Sáez Comet L, Freire Dapena M, Mesa Navas M, Martín Cascón M, et al. Value of nailfold capillaroscopy in the classification of the systemic sclerosis pattern. Med Clin (Barc). 2026;166:107426. doi: 10.1016/j.medcli.2026.107426. PMID: 42013567.