Introduction to nailfold capillaroscopy
Nailfold capillaroscopy is the reference morphological technique for studying distal microcirculation in vivo in patients with Raynaud's phenomenon and suspected connective tissue disease. When properly standardized, it provides clinically relevant diagnostic and prognostic information [1, 2].
- The nailfold window makes it possible to view the capillary loop longitudinally, which is difficult to achieve in most other skin sites [1, 2].
- Its highest clinical yield is in Raynaud's phenomenon, systemic sclerosis, and the scleroderma-spectrum microangiopathies [1, 2, 3, 4].
- The technique is safe, noninvasive, and relatively inexpensive, but its yield depends on standardization, image quality, and reader expertise [1, 2, 6].
- Findings are especially specific when a well-defined scleroderma pattern is present, although this pattern is not exclusive to systemic sclerosis [1, 5, 11].
- Automated artificial intelligence systems are promising, but at present they should be regarded as decision-support tools rather than replacements for expert readers [9, 10].
Learning path
Start here if you need to place the technique, its terminology, and its limits in context. Next, it is useful to review nailfold microcirculation, continue with classical semi-quantitative and quantitative interpretation, and then move on to equipment and devices and acquisition and quality. Later, you can proceed to semiology and reporting and indications and clinical utility when you want to apply the technique to specific clinical questions.
Definition and scope
Before interpreting an image, it is essential to define precisely what capillaroscopy assesses, what it does not assess, and why the nailfold region is used as the standard anatomical window.
What capillaroscopy actually is
Nailfold capillaroscopy, also called nailfold videocapillaroscopy, is the magnified visualization of distal microcirculation in the nailfold region. Its main purpose is to describe capillary morphology and detect signs of structural microangiopathy; it is not a global hemodynamic test and should not be confused with Doppler ultrasound, thermography, or OCT, which answer different physiological questions [1, 2].
In these fundamentals we will preferentially use the terms giant capillary and abnormal shapes, aligned with recent reviews and with the simplified vocabulary now used more often in teaching and classification. In the classical literature, and in some current studies, you will still see megacapillary and branched capillary or ramification; in practice, these usually refer to the same morphological domains using older nomenclature [1, 2].
| Technique | What it adds | Main strength | Important limitation |
|---|---|---|---|
| Nailfold videocapillaroscopy | Capillary morphology, density, hemorrhages, abnormal shapes | High utility in Raynaud's phenomenon and systemic sclerosis | Does not quantify global tissue blood flow by itself [1, 2] |
| Dermoscopy or low-cost digital microscopy | Initial visual screening | Accessibility and speed | Lower resolution and less ability for fine quantification [1, 2] |
| Thermography, Doppler, laser techniques, OCT | Perfusion and functional response | Complementary physiological information | Does not replace standardized morphological analysis [1, 2] |
Why the nailfold region is the window of choice
In the distal nailfold region, the capillary loop runs almost parallel to the skin surface, allowing the entire capillary to be seen along its length rather than only as a point. This anatomical arrangement makes the nailfold region the most useful window for studying capillary density, diameter, and loop architecture [1, 2].
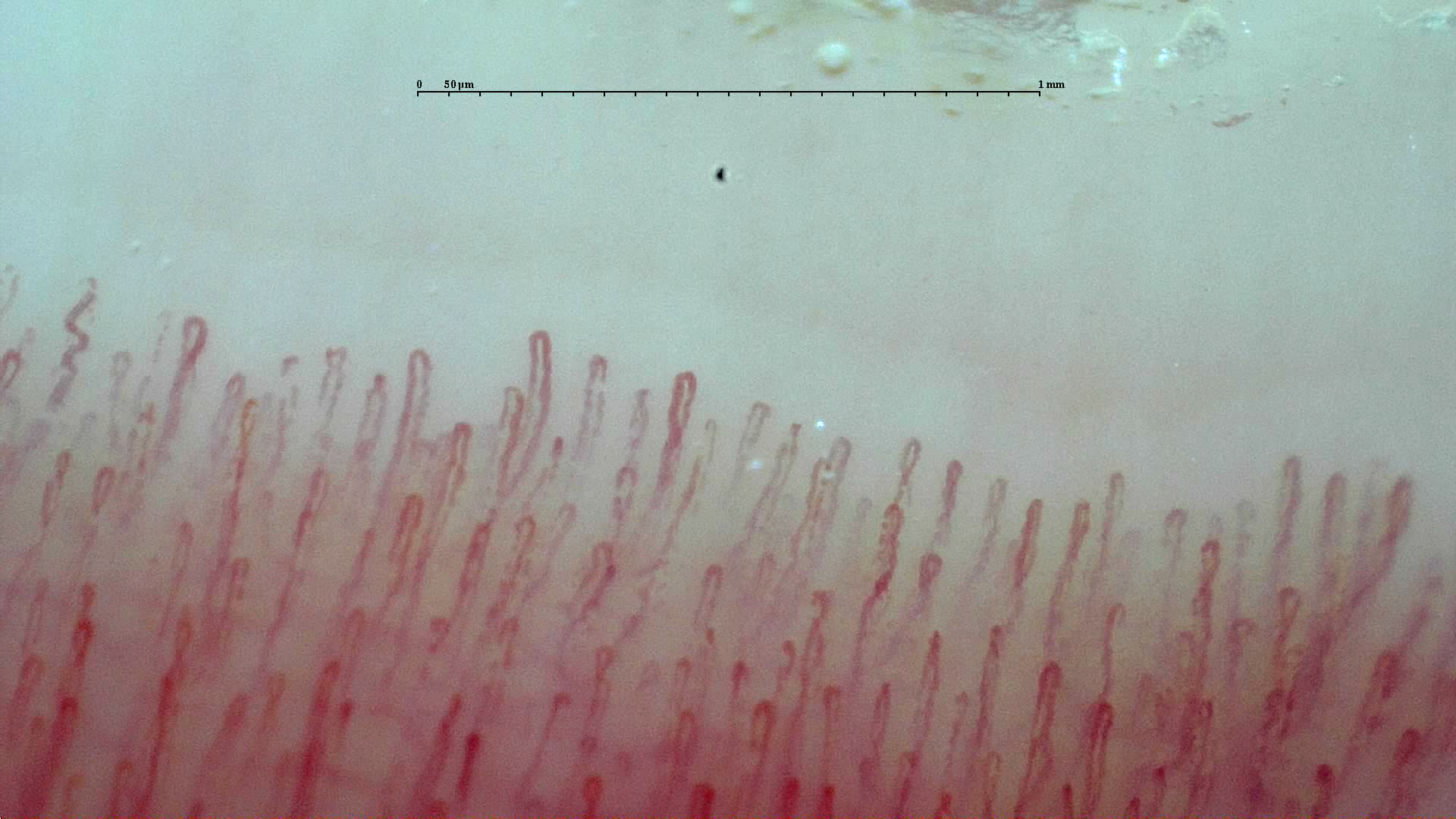
Current clinical role
The main indication is the assessment of Raynaud's phenomenon. In that context, capillaroscopy helps identify structural microangiopathy compatible with connective tissue disease and has been incorporated into the 2013 ACR/EULAR classification criteria for systemic sclerosis [1, 3]. In early-detection programs such as VEDOSS, the combination of abnormal capillaroscopy with autoantibodies and early clinical features allows risk of progression to be stratified [4].
Essential limitations
- Specificity is high for the scleroderma pattern, but low for isolated or nonspecific changes [1, 5, 11].
- Visibility decreases when image quality is poor or when skin phototype, trauma, recent manicure, or excessive device pressure interfere [1, 2].
- There are no universal, self-sufficient criteria that can separate primary from secondary Raynaud's phenomenon on their own in every clinical scenario [1, 11].
Strong evidence: utility in Raynaud's phenomenon and systemic sclerosis. Moderate evidence: early prognostic stratification within the scleroderma spectrum. Insufficient evidence: using capillaroscopy as a stand-alone test to classify any acrosyndrome outside a well-defined clinical context [1, 4, 11].
History and standardization
The technique moved from descriptive microscopic observation into clinical algorithms because it became more reproducible, more comparable between centers, and easier to document.
Brief historical perspective
The development of capillaroscopy followed the evolution of microscopy. Lombard described the distribution of the human nailfold capillary bed in 1912, and Brown and O'Leary published one of the first descriptions of the cutaneous scleroderma pattern in 1925 [12, 13]. Modern digital videocapillaroscopy later consolidated quantification, image archiving, and longitudinal comparison [1, 2].
| Milestone | Practical importance |
|---|---|
| 1912: Lombard describes the nailfold capillary bed [12] | Establishes the nailfold region as a useful anatomical window |
| 1925: Brown and O'Leary describe abnormal capillaries in scleroderma [13] | Links capillary morphology with systemic disease |
| Subsequent decades: video, digital archiving, and software [1, 2] | Facilitates follow-up, quantification, and multicenter studies |
| 2013 onward: integration into classification and early diagnosis [3, 4] | Moves from descriptive tool to clinical stratification component |
| 2019: AI applied to capillaroscopy [17] | Initiates quantitative analysis of the whole nailfold bed, allowing greater objectivity |
From expert observation to standardization
The most cited methodological reviews emphasize that the technique is comparable between centers only when equipment, examined fingers, thermal environment, recorded parameters, and reporting style are standardized [1, 2, 8]. In parallel, Dinsdale's work showed that limiting the examination to fewer fingers reduces sensitivity: compared with the eight-finger standard, examining only four middle or ring fingers reduced sensitivity to 66.7%, and examining only both ring fingers reduced it to 59.8% [7]. A 2026 multicenter study extended this message to nailbed coverage: reducing a 32-image examination of the eight non-thumb fingers to fewer or only medial fields caused substantial disagreement in systemic sclerosis pattern assignment [20].
In 2024, a minimum set of domains for capillaroscopy reporting was proposed to harmonize daily practice and research. The value of this framework is pragmatic: it requires documentation of what was observed, in how many fingers, at what image quality, and using which language [8].
Predictive value: strong with a clear pattern, limited without one
In the VEDOSS cohort, 52.4% of patients with Raynaud's phenomenon and very early features progressed to systemic sclerosis over five years; risk was much higher when disease-specific antibodies, positive ANA, and puffy fingers coexisted, reaching 94.1% in the subgroup with that combination [4]. This supports the prognostic value of capillaroscopy within a multimodal model, not as an isolated marker.
Conversely, a prospective cohort published in 2024 was unable to build capillaroscopic scores with sufficient discriminatory ability to differentiate primary from secondary Raynaud's phenomenon on their own or to predict progression to systemic sclerosis. This finding does not invalidate the technique, but it does reinforce that clinical and serological context remains essential [11].
The evidence is notably weaker in diseases with microvascular involvement outside scleroderma. Recent studies suggest the presence of "different but also specific" abnormalities in other diseases, including inflammatory myopathy, sarcoidosis, diabetes mellitus, and COVID-19 [14, 15, 16].
Reproducibility and automation
The next step is to understand which parameters are more reproducible and how to interpret the rise of artificial intelligence without promising more than the evidence currently supports.
What works best between observers
Reproducibility is not homogeneous across all findings. In a multicenter study with 10 experts and 173 participants, interobserver reliability, conditional on the image being assessable, was high for the global grade (ICC 0.78), capillary density (ICC 0.64), and apical width (ICC 0.85); the main problem was image assessability rather than the measurement itself [6]. Previous studies also showed better agreement for robust quantitative variables than for more subjective morphological features, especially tortuosity or "bizarre" shapes [6].
| Parameter | Current robustness | Practical comment |
|---|---|---|
| Capillary density | High utility and good reproducibility if the image is assessable [2, 6] | Usually more stable than purely qualitative features |
| Giant capillaries and hemorrhages | Good agreement when image quality is adequate [2, 6] | Highly informative within the scleroderma pattern |
| Tortuosity, crossing, or bizarre shapes | Lower reader agreement [6] | Should not be overemphasized without associated findings |
Artificial intelligence and automated analysis
Automation has moved from conceptual promise to measurable results. A vision transformer model trained on 17,126 images from EUSTAR and VEDOSS registries achieved AUC values of 81.8%-84.5% for different microangiopathic abnormalities and an AUC of 88.6% for the scleroderma pattern in a validation subset with consensus labels [9]. In 2025, a deep-learning study on videocapillaroscopy image classification reported 90.6%-98.9% accuracy and 99%-100% AUC in a balanced single-center dataset [10].
These numbers are encouraging, but they must be contextualized: most models are trained with curated databases, selected populations, and image standards that are not always reproduced in routine practice. In addition, results often depend on prior expert consensus, which remains the reference standard [8, 9, 10].
More robustly, the Capillary.io working group has conducted several studies implementing AI to objectify capillaroscopy. This began with an internal validation study, reinforced by a second published study demonstrating applicability in other cohorts (external validity). Finally, the group first worked on objectification and subsequently on automated pattern recognition using AI [17, 18, 19].
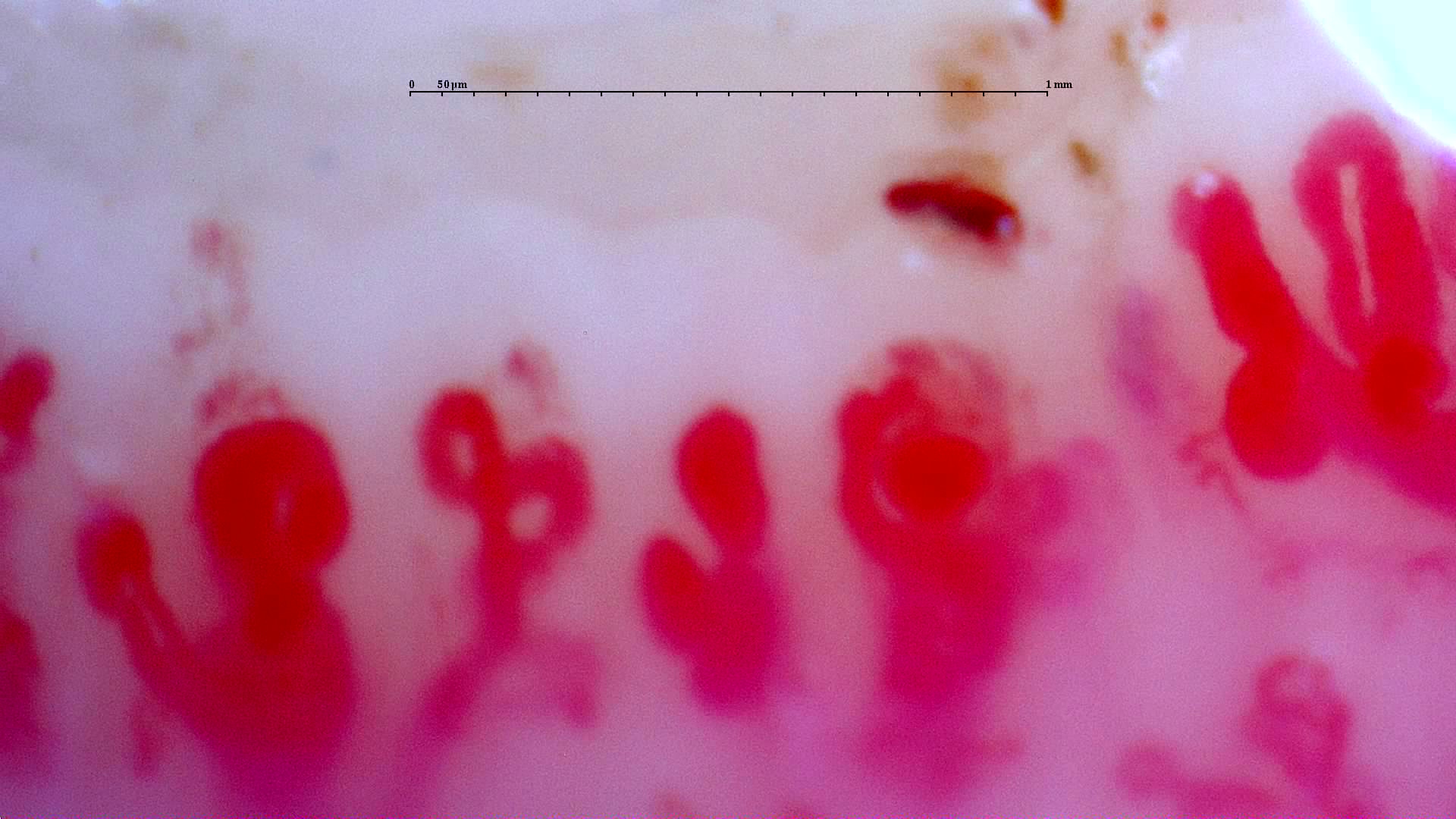
Brief checklist for a rigorous introduction to the technique
- Confirm that the purpose of the test is morphological rather than functional.
- State clearly whether the main clinical suspicion is primary Raynaud's, secondary Raynaud's, or a scleroderma-spectrum disorder.
- Ideally examine eight fingers, excluding thumbs, with systematic medial and lateral nailbed coverage unless a technical limitation is justified [2, 7, 20].
- Record image quality and limit conclusions when assessability is low [6, 8].
- Do not interpret capillaroscopy outside the context of clinical features, autoantibodies, and disease evolution [1, 4, 11].
FAQ
Are capillaroscopy and videocapillaroscopy the same?
Not exactly. "Capillaroscopy" is the umbrella term; in current clinical practice, nailfold videocapillaroscopy is the highest-resolution modality and the one most often used for documentation and follow-up [1, 2].
Does the test hurt, or does it have contraindications?
It is a noninvasive examination and is generally well tolerated. The main practical problems are not safety issues but image quality and interpretation [1, 2].
Does a normal capillaroscopy exclude systemic sclerosis?
Not absolutely. It reduces the probability of overt scleroderma-spectrum microangiopathy at that moment, but it does not replace clinical or serological assessment, especially in very early phases [1, 4, 11].
Is it enough to examine two or four fingers?
It may be useful as a quick screen, but it loses sensitivity and may underclassify systemic sclerosis patterns. The available evidence supports examining eight fingers, with adequate nailbed coverage, unless limitations are justified [2, 7, 20].
Which finding carries the most clinical weight?
The combination of giant capillaries, hemorrhages, density loss, and architectural disorganization, in other words a coherent scleroderma pattern, carries more weight than an isolated abnormality [1, 3, 5].
Can AI already replace an expert reader?
No. It can accelerate and objectify specific tasks, but current evidence places it as support for reading, not as a full substitute for expert interpretation [9, 10].
Glossary
- Nailfold capillaroscopy
- Magnified visualization of nailfold microcirculation to describe capillary morphology.
- Scleroderma pattern
- A set of findings compatible with systemic sclerosis microangiopathy, typically including giant capillaries, hemorrhages, density loss, and neoangiogenesis.
- Giant capillary
- A homogeneously dilated capillary; this is the preferred term in many recent texts and corresponds to the historical term megacapillary. In videocapillaroscopy, it is usually reserved for apical diameters of 50 μm or more, whereas smaller dilations are usually described above 20 μm.
- Abnormal shape
- Umbrella term for irregular capillary morphologies. In the classical literature, it often includes the older terms branched capillaries or ramifications.
- Assessability
- The degree to which an image allows a given parameter to be measured or classified reliably.
- VEDOSS
- Very Early Diagnosis of Systemic Sclerosis, a strategy designed to identify very early scleroderma-spectrum disease.
References
- Smith V, Ickinger C, Hysa E, Snow M, Frech T, Sulli A, et al. Nailfold capillaroscopy. Best Pract Res Clin Rheumatol. 2023;37(1):101849. doi: 10.1016/j.berh.2023.101849. PMID: 37419757.
- Smith V, Herrick AL, Ingegnoli F, Damjanov N, De Angelis R, Denton CP, et al. Standardisation of nailfold capillaroscopy for the assessment of patients with Raynaud's phenomenon and systemic sclerosis. Autoimmun Rev. 2020;19(3):102458. doi: 10.1016/j.autrev.2020.102458. PMID: 31927087.
- van den Hoogen F, Khanna D, Fransen J, Johnson SR, Baron M, Tyndall A, et al. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2013;65(11):2737-2747. doi: 10.1002/art.38098. PMID: 24122180.
- Bellando-Randone S, Del Galdo F, Lepri G, Czirják L, Denton CP, Guiducci S, et al. Progression of patients with Raynaud's phenomenon to systemic sclerosis: a five-year analysis of the European Scleroderma Trial and Research group multicentre, longitudinal registry study for Very Early Diagnosis of Systemic Sclerosis (VEDOSS). Lancet Rheumatol. 2021;3(12):e834-e843. doi: 10.1016/S2665-9913(21)00244-7. PMID: 38287630.
- Roberts-Thomson PJ, Patterson KA, Walker JG. Clinical utility of nailfold capillaroscopy. Intern Med J. 2023;53(5):671-679. doi: 10.1111/imj.15966. PMID: 36353844.
- Dinsdale G, Moore T, O'Leary N, Tresadern P, Berks M, Roberts C, et al. Intra- and inter-observer reliability of nailfold videocapillaroscopy: a possible outcome measure for systemic sclerosis-related microangiopathy. Microvasc Res. 2017;112:1-6. doi: 10.1016/j.mvr.2017.02.001. PMID: 28163035.
- Dinsdale G, Roberts C, Moore T, Manning J, Berks M, Allen J, et al. Nailfold capillaroscopy: how many fingers should be examined to detect abnormality? Rheumatology (Oxford). 2019;58(2):284-288. doi: 10.1093/rheumatology/key293. PMID: 30247696.
- El Miedany Y, Ismail S, Wadie M, Müller-Ladner U, Giacomelli R, Liakouli V, et al. Development of a core domain set for nailfold capillaroscopy reporting. Reumatol Clin (Engl Ed). 2024;20(7):345-352. doi: 10.1016/j.reumae.2024.07.003. PMID: 39160005.
- Garaiman A, Nooralahzadeh F, Mihai C, Perez Gonzalez N, Gkikopoulos N, Becker MO, et al. Vision transformer assisting rheumatologists in screening for capillaroscopy changes in systemic sclerosis: an artificial intelligence model. Rheumatology (Oxford). 2023;62(7):2492-2500. doi: 10.1093/rheumatology/keac541. PMID: 36347487.
- Yayla ME, Aydın A, Kılıçaslan M, Kalkan M, Güzel MS, Shikhaliyeva A, et al. Deep learning performance in analyzing nailfold videocapillaroscopy images in systemic sclerosis. Diagnostics (Basel). 2025;15(22):2912. doi: 10.3390/diagnostics15222912. PMID: 41300936.
- Amaral MC, Paula FS, Caetano J, Ames PR, Alves JD. Re-evaluation of nailfold capillaroscopy in discriminating primary from secondary Raynaud's phenomenon and in predicting systemic sclerosis: a randomised observational prospective cohort study. Expert Rev Clin Immunol. 2024;20(6):665-672. doi: 10.1080/1744666X.2024.2313642. PMID: 38465507.
- Lombard WP. The blood pressure in the arterioles, capillaries and small veins of the human skin. Am J Physiol. 1912;29:335-362.
- Brown GE, O'Leary PA. Skin capillaries in scleroderma. Arch Intern Med (Chic). 1925;36(1):73-88. doi: 10.1001/archinte.1925.00120130076008.
- Mondini L, Confalonieri P, Pozzan R, Ruggero L, Trotta L, Lerda S, Hughes M, Bellan M, Confalonieri M, Ruaro B, Salton F, Tavano S. Microvascular Alteration in COVID-19 Documented by Nailfold Capillaroscopy. Diagnostics (Basel). 2023;13(11):1905. doi: 10.3390/diagnostics13111905. PMID: 37296759.
- Chianese M, Screm G, Confalonieri P, Salton F, Trotta L, Da Re B, Romallo A, Galantino A, D'Oria M, Hughes M, Bandini G, Confalonieri M, Baratella E, Mondini L, Ruaro B. Nailfold Video-Capillaroscopy in Sarcoidosis: New Perspectives and Challenges. Tomography. 2024;10(10):1547-1563. doi: 10.3390/tomography10100114. PMID: 39453031.
- Shah R, Petch J, Nelson W, Roth K, Noseworthy MD, Ghassemi M, Gerstein HC. Nailfold capillaroscopy and deep learning in diabetes. J Diabetes. 2023;15(2):145-151. doi: 10.1111/1753-0407.13354. PMID: 36641812.
- Gracia Tello B, Ramos Ibañez E, Fanlo Mateo P, Sáez Cómet L, Martínez Robles E, Ríos Blanco JJ, Marí Alfonso B, Espinosa Garriga G, Todolí Parra J, Ortego Centeno N, Callejas Rubio JL, Freire Dapena M, Marín Ballvé A, Selva-O'Callaghan A, Guillén Del Castillo A, Simeón Aznar CP, Fonollosa Pla V. The challenge of comprehensive nailfold videocapillaroscopy practice: a further contribution. Clin Exp Rheumatol. 2022;40(10):1926-1932. doi: 10.55563/clinexprheumatol/6usce8. PMID: 34936544.
- Gracia Tello BDC, Sáez Comet L, Lledó G, Freire Dapena M, Mesa MA, Martín-Cascón M, Guillén Del Castillo A, Martínez Robles E, Simeón-Aznar CP, Todolí Parra JA, Varela DC, Maldonado Vélez G, Marín Ballvé A, Aramburu Llorente J, Pérez Abad L, Ramos Ibáñez E. Capi-score: a quantitative algorithm for identifying disease patterns in nailfold videocapillaroscopy. Rheumatology (Oxford). 2024;63(12):3315-3321. doi: 10.1093/rheumatology/keae197. PMID: 38530791.
- Lledó-Ibáñez GM, Sáez Comet L, Freire Dapena M, Mesa Navas M, Martín Cascón M, Guillén Del Castillo A, Simeon CP, Martinez Robles E, Todolí Parra J, Varela DC, Maldonado G, Marín A, Pérez Abad L, Aramburu J, Vela L, Ramos Ibáñez E, Del Carmelo Gracia Tello B. CAPI-Detect: machine learning in capillaroscopy reveals new variables influencing diagnosis. Rheumatology (Oxford). 2025;64(6):3667-3675. doi: 10.1093/rheumatology/keaf073. PMID: 39918978.
- Guillén del Castillo A, Lledó-Ibáñez GM, Sáez Comet L, Freire Dapena M, Mesa Navas M, Martín Cascón M, et al. Value of nailfold capillaroscopy in the classification of the systemic sclerosis pattern. Med Clin (Barc). 2026;166:107426. doi: 10.1016/j.medcli.2026.107426. PMID: 42013567.